
CAPABILTIES
Modern Technology for Modern Research
ChemSCAN brings efficiency and flexibility to catalytic research with features legacy systems simply can’t match.
SPECIFICATIONS
Built for Demanding Research
From early-stage screening with volumes as low as 3 mL to larger scale development, ChemSCAN adapts to your needs. Material-limited? No problem.
| Typical Applications | Catalyst screening (hydrogenation), process optimization, scale-up |
| Vessel Material | 316 stainless steel, Hastelloy C276 |
| Volume Options | 16 mL, 25 mL, 50 mL, 75 mL, 150 mL, 300 mL, 500 mL |
| Working Volume | 3 mL to 400 mL (reactor dependent) |
| Temperature Range | -40°C to 200°C |
| Pressure Range | Standard 100 bar, optional |
TESTIMONIALS
What our Customers Say
Real feedback from industry leaders
Industries and Applications
Hydrogen economy for industrial and energy applications
Development of catalytic processes for hydrogen production from fossil fuels
Industrial paints, alcohols / Oxo-Process -Aldehyde synthesis
Perform oxo Process, hydroformylation, and carbonylation of Aldehydes.
Syngas & CO-Based Catalysis
Investegate reactions involving CO, CO₂, syngas, and mixed gas feeds under regulated pressure for carbonylation,…
Pharma, Fine Chemicals, Food/Cooling Crystallization
Control cooling profiles to optimise nucleation and growth, achieving desired crystal size and purity.
Production of liquid medication into tablet form
Research into solubility of liquids to determine the temperature range over which suitable tablets can…
Separation of chemicals
Crystallization of selected chemicals in order to separate them (by filtration) from other more soluble…
Seeded Crystallization
Use seed crystals to control growth kinetics and reduce batch variability for consistent product quality.
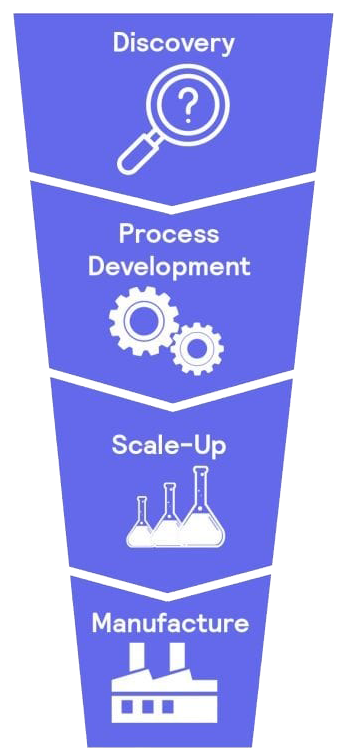
Process safety workflow
Discovery – Chemical Reaction Route Selection & Thermal Screening
Thermal screening helps identify the safest and most efficient route for synthesizing your target molecule. By evaluating safety, complexity, and cost, we ensure that decisions made early in the process minimize risks.
Key Parameters to Obtain During Thermal Screening:
- Onset temperature of decomposition (Td)
- Rate of temperature increase (dT/dt and d²T/dt²)
- Rate of pressure increase (dP/dt)
What our Customers Say

My experience with H.E.L dates since 2005. During these years H.E.L was the provider of the most important lab equipment with the help of which I developed many different API pharmaceutical processes in the companies l worked for including Merck, Pfizer, and Theravance. The versatility of the automated lab reactors (AutoLAB), the ease of use of and the accuracy of the TSu and Simular systems, as well as the flexibility of the PolyBLOCK made my work much easier and accurate. I would like to mention the great relationship I established with the service engineers and the sales people at H.E.L. With their help I was able to customize the software and the hardware of the equipment to our specific needs. This readiness to accommodate each customer’s specific needs gives H.E.L the edge over their competitors and makes them the preferred vendor for pharmaceutical lab equipment.
Process Development and Optimization
At this stage, the focus is on optimizing chemical routes to improve yield, quality, and safety while reducing hazards. Safety is integrated early to establish a strong foundation for small-scale production.
Key safety factors include:
- Reactant accumulation & heat evolution
- Heat capacity & adiabatic temperature rise
- Maximum Temperature of Synthesis Reaction (MTSR)
- Cooling requirements
Safe handling and storage of materials
A key aspect of process development is also ensuring the safe handling and storage of materials. Thermally unstable substances can react at certain temperatures, leading to risks like decomposition, pressure buildup, or explosions. Evaluating thermal stability helps define safe storage conditions and minimize hazards.
What our Customers Say

My experience with H.E.L dates since 2005. During these years H.E.L was the provider of the most important lab equipment with the help of which I developed many different API pharmaceutical processes in the companies l worked for including Merck, Pfizer, and Theravance. The versatility of the automated lab reactors (AutoLAB), the ease of use of and the accuracy of the TSu and Simular systems, as well as the flexibility of the PolyBLOCK made my work much easier and accurate. I would like to mention the great relationship I established with the service engineers and the sales people at H.E.L. With their help I was able to customize the software and the hardware of the equipment to our specific needs. This readiness to accommodate each customer’s specific needs gives H.E.L the edge over their competitors and makes them the preferred vendor for pharmaceutical lab equipment.
Process Scale-Up & Hazard Mitigation
Scaling up a chemical process requires understanding potential hazards. A Process Hazard Analysis (PHA) identifies risks and anticipates worst-case scenarios to ensure safe operations. Key methods include HAZOP, FMEA, Fault Tree Analysis, and Batch Sheet Review.
Adiabatic calorimetry (Phi-TEC I & II) simulates worst-case thermal scenarios, ensuring accurate heat retention and industrial conditions.
Key insights from testing include:
- Tempering Characteristics – Can vapors control heat?
- Discharge Nature – Will the reaction release gas only or a liquid-gas mix?
- These insights are essential for accurate vent sizing and relief system design.
What our Customers Say
No Testimonials found.
Discovery – Chemical Reaction Route Selection & Thermal Screening
Thermal screening helps identify the safest and most efficient route for synthesizing your target molecule. By evaluating safety, complexity, and cost, we ensure that decisions made early in the process minimize risks.
Key Parameters to Obtain During Thermal Screening:
- Onset temperature of decomposition (Td)
- Rate of temperature increase (dT/dt and d²T/dt²)
- Rate of pressure increase (dP/dt)
Process Development and Optimization
At this stage, the focus is on optimizing chemical routes to improve yield, quality, and safety while reducing hazards. Safety is integrated early to establish a strong foundation for small-scale production.
Key safety factors include:
- Reactant accumulation & heat evolution
- Heat capacity & adiabatic temperature rise
- Maximum Temperature of Synthesis Reaction (MTSR)
- Cooling requirements
Safe handling and storage of materials
A key aspect of process development is also ensuring the safe handling and storage of materials. Thermally unstable substances can react at certain temperatures, leading to risks like decomposition, pressure buildup, or explosions. Evaluating thermal stability helps define safe storage conditions and minimize hazards.
Process Scale-Up & Hazard Mitigation
Scaling up a chemical process requires understanding potential hazards. A Process Hazard Analysis (PHA) identifies risks and anticipates worst-case scenarios to ensure safe operations. Key methods include HAZOP, FMEA, Fault Tree Analysis, and Batch Sheet Review.
Adiabatic calorimetry (Phi-TEC I & II) simulates worst-case thermal scenarios, ensuring accurate heat retention and industrial conditions.
Key insights from testing include:
- Tempering Characteristics – Can vapors control heat?
- Discharge Nature – Will the reaction release gas only or a liquid-gas mix?
- These insights are essential for accurate vent sizing and relief system design.












