Quality Control
How can calorimetry help that cells pass quality control?
As the use of lithium-ion batteries continues to grow in popularity and importance, both in industry, in transport, and in day-to-day activities, it is important that these battery are safe and reliable. In the months running to this post, an increase number of accidents involving Lithium-ion batteries have been reported in the press. This highlights the importance of robust testing and strong legislation to ensure the safety of users.
The United Nations (UN) established the Manual of Tests and Criteria (UN 38.3) to set standards for the safe transportation of batteries. This manual outlines the tests and requirements that batteries must meet in order to be considered safe for transportation. The International Electrotechnical Commision (IEC), through the IEC 62133 also established what that the safety standards are for batteries in consumer electronics. Amongst these standards, it outlines what the requirements are to cover electrical, mechanical, and environmental terms. Similarly, the Underwriters Laboratories (UL) published UL2054, establishing the safety standards in various applications, including consumer electronics, energy storage, and automotive.
Other regulatory bodies that have published safety guidelines on battery safety include the International Organization for Standardization (ISO), which published the ISO 12405, covering the requirements for batteries for electric vehicles regarding electrical, mechanical, and environmental safety. The Japanese Industrial Standards (JIS) through JIS C 8714 and the Chinese National Standard (GB/T) also cover the same aspects of batteries. In the US, the UL 1642, although not legally binding, contains the recommendations for lithium-containing batteries and focuses on their thermal, mechanical, and electrical characteristics and fire and shock resistance.
So, we can see a growing body of internal legislation that aims to produce safer batteries.
Commonalities
Legislation, directives, and battery recommendations aim to produce safer batteries, but what is considered safe?
On the electrical side, batteries must withstand potential stress, including overcharging and over-discharging, without the potential of a fire or explosion hazard. Overcharging occurs due to excessive voltage, causing current to flow into the battery after reaching full charge. Overdischarging, on the other hand, is the discharging of the battery beyond the lower safe voltage or discharging a battery at an unsuitable current. The result of both processes is the overheating of batteries, which can result in the generation of thermal runaways.
Mechanically, batteries should be able to endure various mechanical stresses, such as impact, vibration, or crush tests, without rupturing, leaking, or short circuits (both internal and external).
Battery operation sometimes includes transient conditions that can affect batteries. These include high temperatures (which can result in thermal runaways), low temperatures (reduce the ion transmission capacity of the electrolyte), humidity, or different atmospheric pressures.
As we can see, batteries can result in temperature anomalies due to their own functioning or environmental conditions. Batteries need to be able to dissipate heat to maintain safe operation temperatures, therefore preventing hazardous scenarios.
Battery testing
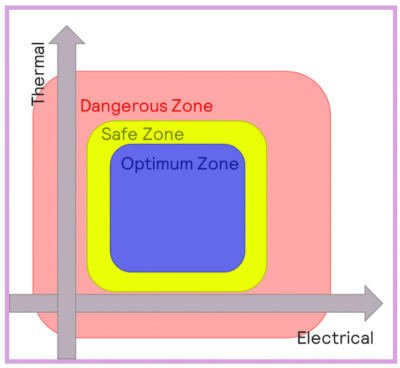
Batteries will behave differently when operating at different temperatures, and three main thermal regions can be defined:
- Optimum zone: batteries operating within optimal conditions will behave normally, and aging processes will be as slow as possible.
- Safe zone: the battery will function in this zone, but performance can be compromised, especially charging/discharging speed and aging. Higher temperatures can result in accelerated degradation of the electrolyte. Lower temperatures can result in a reduction in the conductivity of the electrolyte, as well as the deposition of lithium-organic compounds onto the SEI.
- Dangerous zone: conditions are likely to result in hazardous scenarios.
Calorimetry is an invaluable tool for the testing of batteries.
Isothermal calorimeters maintain the battery at a constant temperature and measures its thermal energy changes. It is possible to monitor the electrical output of the battery, thus delivering both thermal and electrical behaviour information. Integrating the instrument with a charge-discharge unit enables the automation of repeated cycling of battery cells under various charge and discharge rates. This type of testing allows for determining the battery’s performance within the Optimum Zone and Safe Zone.
On the other hand, Adiabatic calorimeters maintain batteries under adiabatic conditions, where there is no heat exchange with the surroundings. This calorimeter allows us to subject the battery to extreme conditions within a safe environment to evaluate the consequences. These conditions include higher temperatures, voltages, and discharge currents outside the Safe Zone. Some adiabatic calorimeters can be equipped with means to test for mechanical stress, such as crushing or perforation. Adiabatic calorimetry enables the battery response within the Dangerous Zone to be explored.
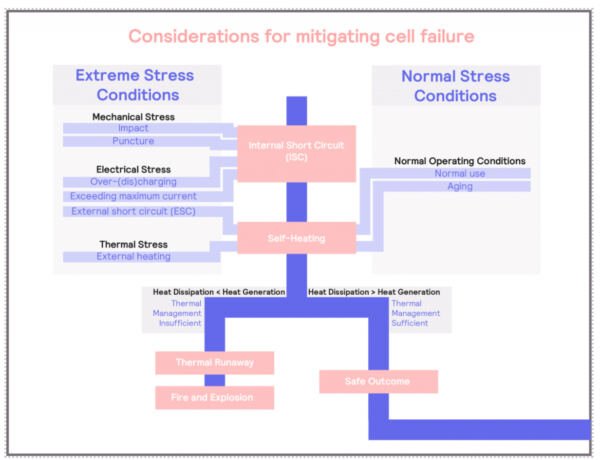
Considerations for Mitigating Cell Failure
Solutions
The iso-BTC is designed as an isothermal calorimeter, allowing battery performance testing. Battery efficiency and heat evolved at specific temperatures, and charge-discharge rates are fundamental parameters that must be tested for cell manufacturers and integrators. Moreover, identifying safe operating limits to avert the risk of thermal runaway is fundamental. Batteries must be tested to obtain a safe working temperature, maximum safe voltage, and maximum safe current. These values can be obtained using BTC-130 and BTC-500. These battery operational parameters need to be appropriate and comply with the current legislation in order for the batteries and battery-powered devices to be commercialized.

iso-BTC | Bench-top, battery performance testing, isothermal calorimeter
The iso-BTC (Battery Testing Calorimeter) is an isothermal calorimeter designed for the ch...

BTC-130 | Bench-Top, Battery Safety Testing, Adiabatic Calorimeter
The BTC-130 (Battery Testing Calorimeter) is a bench-scale adiabatic calorimeter designed ...

BTC-500 | Large-scale, battery testing, adiabatic calorimeter
The BTC-500 (Battery Testing Calorimeter) is a floor-standing adiabatic calorimeter for th...



