Does today’s take-up of flow chemistry in fields such as photochemistry, electrochemistry, and hazardous gas usage mean that the days are numbered for parallel batch reactor testing? We’ve been talking to leading experts in flow catalysis in pharmaceutical companies to determine how they address this topic.
- Batch and Flow – the basics
- What’s the Pharma point of view?
- Batch in Pharma
- Going with the Flow in Pharma
- Key advantages of Flow Hydrogenation
- The ideal zone for Pharma catalysts
Batch and flow – the basics
We’re all familiar with parallel batch reactor systems that allow for several catalyzed reactions to undergo fast and efficient screening simultaneously. Starting materials are loaded, reacted, and then analyzed. Only at the end of the reaction can the full impact of experimental variables be seen. Sampling is possible during the reaction to see how the conversion is progressing, but be aware that changing reaction conditions can mislead you into drawing incorrect conclusions. You also need to consider that larger-scale batch reactions need time to charge, heat and cool, or reach working reaction conditions before the reaction of interest occurs. These delays of working at scale can have wider technical and commercial implications.
Things are different with flow chemistry. A small amount of active reactants are under reaction conditions at any one time and are continuously refreshed. This creates a flowing stream of product, making it possible to test the efficiency and effectiveness of the reaction continually. Any change in conditions will have a virtually immediate impact on the reaction and thus the product. Flow has the ability for samples to be taken at sensible points of the elutes flow, so multiple variables and reaction conditions can be investigated in one experiment.
The opportunity of flow chemistry offers five key benefits:
1. Increased safety
2. Greater sustainability
3. Speed
4. Increased flexibility
5. Automation
What’s the Pharma point of view?
In the pharmaceutical industry, catalytic hydrogenation reactions are typically conducted in batch mode. However, these reactions tend to benefit from non-stop processing, as the catalytic (continuous or) flow reactors can offer high catalyst loading, high-pressure operation and enhanced mass transfer on a smaller, more economical scale. Even established pharmaceutical companies require constant improvements in their manufacturing processes. Every gain in efficiency of reaction or improving the speed to market for an API may significantly enhance the company’s profitability. Thus, revaluating the traditional batch methods as flow experiments should be considered in all pharma value chains.
Batch at Pharma
Batch hydrogenation in pharma creates specific, clear challenges. These are generally complex three-phase reactions that have low heat and mass transfer and are sensitive to scale. It requires large volumes of compressed hydrogen, especially in large production size vessels, and this directly limits operating pressure. It’s common that, for safety reasons, batch reactions are limited to low pressures due to the large volumes of hydrogen gas used, generally within the region of 5 to 10 bar. Batch also creates the necessity for catalyst powders to be manipulated in large amounts, including filtration at the end of the reaction. Each additional step will impact the cost-effectiveness of each process.
An additional challenge of batch processing is the length of time it takes to clean the vessels. Depending on the size and process undertaken, it can take up to a week to clean a reactor. This highly manual process is carried out in shifts, and sometimes it’s necessary to climb into the vessel. If these hazardous and time-consuming steps can be removed/avoided, I’m sure everyone who has to clean the reactor would be grateful.
A large number of batch hydrogenation reactions look specifically to:
- Monitor substrates, products, and intermediates over time
- See the effects of operating conditions on yield and kinetics. The key variables usually are concentration, temperatures, pressure, and amount of catalyst loading
- Provide information on reaction mechanisms and to estimate kinetic constraints
Going with the flow in Pharma
Flow chemistry allows you to understand the:
- Conversion and yield data for varying catalyst mass and operating conditions quickly, within one experimental run.
- The effect of Flow rates (Flow regimes) on reaction performance
- The effect that scale will have on the reaction, such as the effect of temperature gradient.

In our recent webinar, Lee Edwards, an investigator and research fellow at GSK, explains the benefits of flow chemistry to GSK, “Simply put, a continuous reactor is continuously reacting! For an operation like ours this allows continuous manufacturing; materials are simultaneously charged and discharged from the process. With it, we achieve more efficient reactor usage, applying smaller reactors that still allow the same throughput as something larger. You’ll also find that flow is far more suitable than batch when working with pyrophoric materials: there are just some things you don’t want to be handling!
We find that we achieve better control of process parameters and see improved product quality controls for it in flow. And the long-term benefit of that is our ability to provide improved quality into the pharma arena.”
Key advantages of flow hydrogenation:
- Safer reactions. Less of the reagent is under hazardous conditions at any one time, leading to inherently less hazardous environments.
- Flow rates control gas distribution and mass/heat transfer
- Allows for high local catalyst loading, enabling difficult transformations
- Smaller equivalent volume allows for higher pressure, supporting new chemistry.
- Close to steady-state operation – helps create better operational control
- Allows a more extensive and varied range of catalysts to be used
- Simpler catalyst handling that avoids the need for filtration.
The ideal zone for Pharma catalysts
For Lee and the team at GSK, finding the right flow catalyst is mainly about particle size. Pharma is generally interested in particle sizes between 50-400 microns and the GSK team currently have 35 catalysts in this range.
By contrast, batch reactions typically need fine powders to traverse from lab to plant, typically around 10 microns. The problem to be aware of here is if there are any pressure drops happening in the reactor. Lee explains, “The pressure drop is the difference in pressure between the top and bottom of the column. So, if you want to run a hydrogenous solution at 10 Bar and had a 20 Bar pressure drop, you’d need 30 Bar of pressure at the top of the column for it to be 10 Bar at the bottom. This makes it (10 micron catalysts) an unsuitable choice for continuous pharma hydrogenations as you just can’t scale properly. You’ll find industrial catalysts very much at the other end of the scale, requiring reactants by the kilo.”
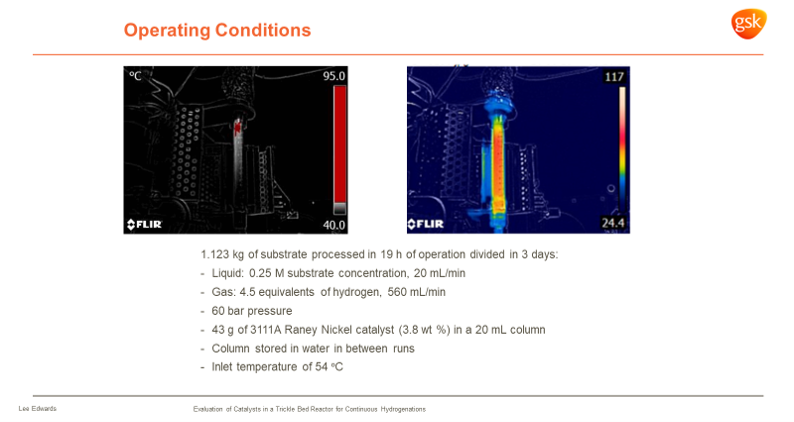 |
Figure 1: Typical FlowCAT Operating Conditions at GSK (Taken from presentation at our H.E.L Flow Chemistry Mini-Symposium 2021)
GSK currently carries out their testing for scale up on H.E.Ls configurable fixed-bed flow reactor, the FlowCAT. They first installed the standard 200 bar/300 oC unit in 2016 and have found the reactor’s configurability an advantageous aspect of it.
If you wish to read more on GSK’s use of the FlowCAT, read our application note covering their work. This work has been published by the ACS, with the full paper, available from the ACS here.
 |
Figure 2: GSK scale up conclusions about the FlowCAT (Taken from presentation at our H.E.L Flow Chemistry Mini-Symposium 2021)
Investigating the use of flow chemistry in pharmaceutical research and development does highlight flow’s many benefits. Not only can flow chemistry allow for API’s synthesis to be investigated and optimized on a smaller scale in a faster time, but it also provides a safer and more economical platform to do so. For more insights into flow chemistry in its many aspects, please watch the full webinar recording of the H.E.L Flow Chemistry Mini-Symposium 2021








