CrystalEYES | Crystallization Monitoring System
CrystalEYES is an advanced crystallization monitoring system designed to measure turbidity, temperature, and optional pH during crystallization processes. It enables real time detection of dissolution and nucleation points, supporting accurate crystallization analysis and process development. With scalable probe options and flexible upgrades, CrystalEYES provides a versatile solution for crystallization monitoring across all laboratory scales.
Overview
Crystallization Monitoring and Analysis
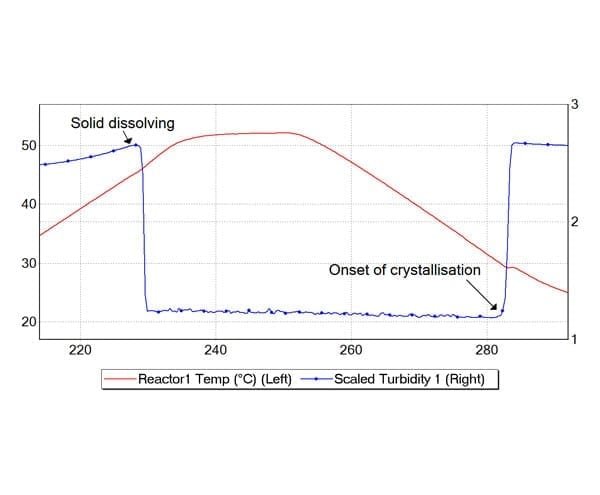
Crystallization plays a critical role in determining the quality, performance, and manufacturability of chemical and pharmaceutical products. CrystalEYES enables chemists to monitor crystallization processes in real time, providing clear identification of dissolution and nucleation events. This supports improved understanding and control of crystallization behavior.
Flexible Measurement Capabilities
The system measures turbidity and temperature as standard, with optional pH monitoring where required. This combination of parameters provides a comprehensive view of crystallization processes, enabling more accurate data collection and analysis.
Scalable Across All Development Stages
CrystalEYES offers a wide range of probe options to support different scales, from small test tubes of around 3 milliliters to large vessels exceeding 10 liters. Probes are available in materials such as 316 stainless steel and Hastelloy, ensuring compatibility with a wide range of chemistries. Scaling between applications is simple, requiring only a probe change without modification to the system electronics or software.
Integrated Data Logging and Analysis
All experimental data is automatically recorded in CSV format, allowing easy transfer to Excel or other analysis tools. This ensures efficient data handling and supports rapid interpretation of crystallization results.
Upgradeable Control and Multi Reactor Monitoring
CrystalEYES can be upgraded to include full process control, enabling precise management of heating, cooling, stirring, and liquid dosing. Multi channel configurations are also available, allowing simultaneous monitoring and control of multiple reactors for increased throughput and efficiency.
Publications
The following are a list of some technical publications which highlight the use of the equipment.
Effects of Various Inhibitors on the Nucleation of Calcium Oxalate in Synthetic Urine
Yu-Chao Hsu, Yi-Hsuan Lin, Lie-Ding Shiau
01-Apr-2020
https://doi.org/10.3390/cryst10040333(Subscription or purchase maybe required for full access)
A practical guide to the measurement of turbidity curves of cooling crystallisations from solution
Simon J. Coles ORCID logo and Terence L. Threlfall
01-Feb-2020
https://doi.org/10.1039/C9CE01622H(Subscription or purchase maybe required for full access)
Determination of Solubility and Nucleation Kinetics of Valnemulin Hydrochloride Solvate
Jinbo Ouyang, Bing Na, Zhirong Liu, Limin Zhou, Hongxun Hao
01-Mar-2019
https://doi.org/10.1007/s10953-019-00861-7(Subscription or purchase maybe required for full access)
The temperature dependence of the pre-exponential factor and interfacial energy for aqueous glycine solutions based on the metastable zone width data
Lie-Ding Shiau
01-May-2018
https://doi.org/10.1016/j.jcrysgro.2018.05.020(Subscription or purchase maybe required for full access)
Solid-liquid equilibrium for proteins in solutions with an unconventional salt (ammonium carbamate): Phase behavior analysis
G. Hirata, P. Filho, E. Miranda
01-Apr-2017
https://doi.org/10.1016/j.fluid.2017.03.031(Subscription or purchase maybe required for full access)
Efficient Solvent Selection Approach for High Solubility of Active Phytochemicals: Application for the Extraction of an Antimalarial Compound from Medicinal Plant
Souhila Laboukhi-Khorsi,†,‡ Kamel Daoud,‡ and Smain Chemat
01-Mar-2017
https://doi.org/10.1021/acssuschemeng.7b00384(Subscription or purchase maybe required for full access)
Effect of silica nanoparticles to prevent calcium carbonate scaling using an in situ turbidimetre
W.N.Al NasseraU.V.ShahbK.NikiforoubP.PetroubJ.Y.Y.Hengb
01-Dec-2015
https://doi.org/10.1016/j.cherd.2015.12.006(Subscription or purchase maybe required for full access)
Metastable Zone Widths, Conformational Multiplicity, and Seeding
Terence L. Threlfall,* Russell W. De’Ath, and Simon J. Coles
01-Feb-2013
https://doi.org/10.1021/op3003486(Subscription or purchase maybe required for full access)

I use a H.E.L. turbidity probe plus temperature probe for the determination of metastable zone widths and latent/induction time measurements in the study of crystallisation behaviour. The system is easy to use and very robust and records faithfully all the happenings. Even if one manages to do silly things, the data is still there and recoverable. The probes are neat and so can be used in small vessels, an advantage when dealing with valuable compounds. The amount of data that the computer will hold from a run is astonishing. The help from the technical team is always there, so we have been very pleased with our purchase.